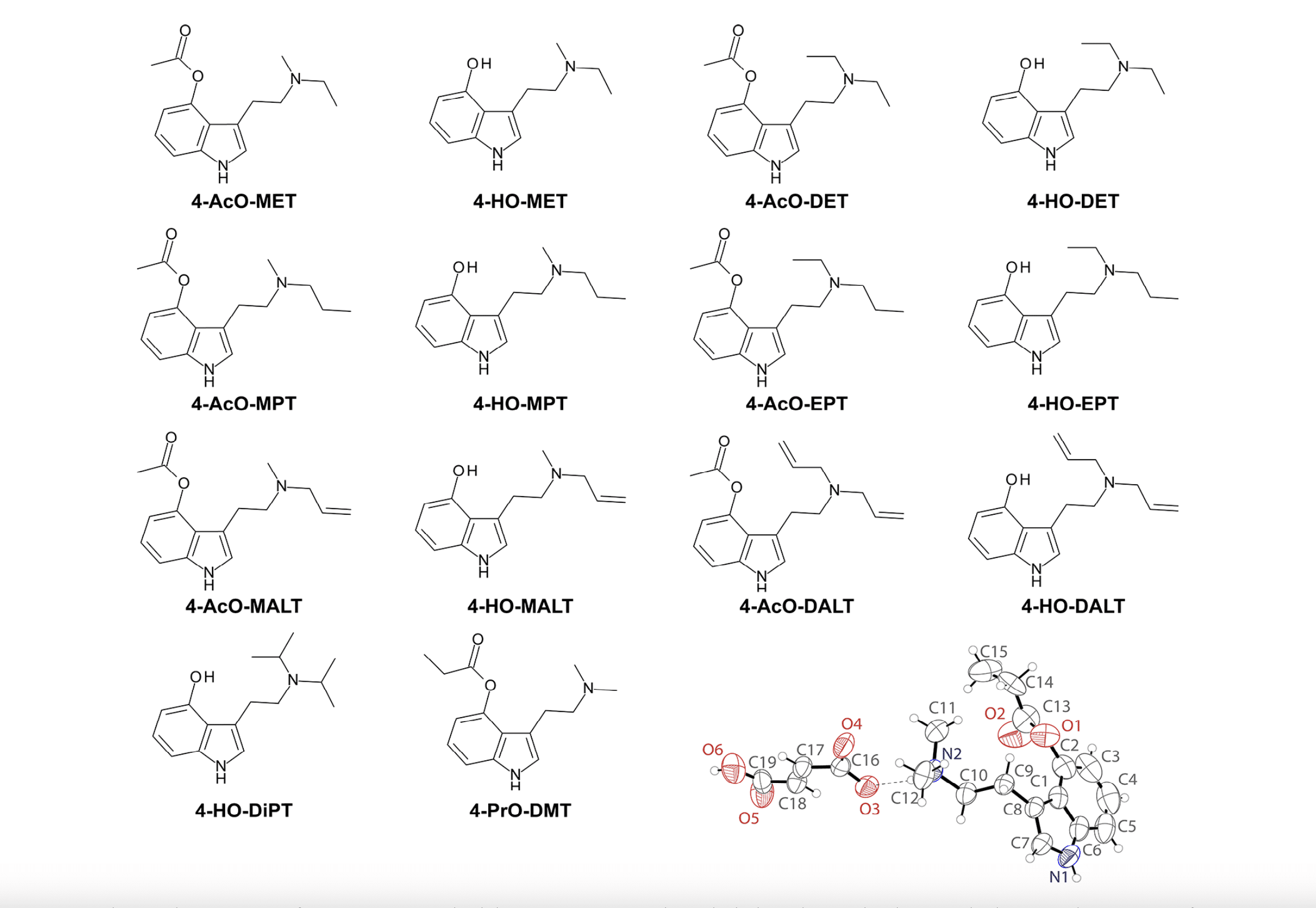
Through its Cooperative Research and Development Agreement with the Designer Drug Research Unit at the National Institute on Drug Abuse (NIDA) Intramural Research Program (part of the National Institutes of Health [NIH]) and in collaboration with scientists from the Manke Lab at UMass Dartmouth, CaaMTech et al. recently published receptor binding profiles for a variety of psychedelic tryptamines and head-twitch response (HTR) data for the synthetic psilocybin analog 4‑propionoxy-N,N-dimethyltryptamine (4-PrO-DMT). “Receptor Binding Profiles for Tryptamine Psychedelics and Effects of 4-Propionoxy-N,N-dimethyltryptamine in Mice” was published in ACS Pharmacology & Translational Science. The new collaborative research progresses CaaMTech’s scientific investigation of novel, synthetic alternatives to psilocybin. Psilocybin has drawn widespread attention and scientific investigation because of its therapeutic potential, but recent scientific investigation into an alternative prodrug of its active metabolite, psilocin, highlights opportunities for improvement.
4-PrO-DMT is a synthetic tryptamine compound closely structurally related to psilocybin (4-PO-DMT). 4-PrO-DMT is purported to be a psilocin (4-HO-DMT) prodrug, though further scientific investigation is needed to verify this hypothesis. Psilocin prodrugs such as psilocybin and psilacetin are metabolized into psilocin, which induces psychedelic effects.
Little is scientifically known about the pharmacology of 4-PrO-DMT. Substances labeled as 4-PrO-DMT have appeared for sale in online gray market “research chemical” markets and people who use these substances have reported subjective experiences that are similar to those produced by psilocin, psilocybin, and psilacetin. The data from this publication represent the first comprehensive preclinical pharmacological investigation of 4-PrO-DMT and fill in blanks regarding differences in potential biological targets of the various psychedelic tryptamines tested. The work specifically determined comparative potential biological target profiles of various tryptamine psychedelics structurally related to psilocybin and measured the psychedelic-like effects of 4-PrO-DMT in mice via HTR assays. Further, the study results add to a growing body of evidence suggesting that the 5-HT1A receptor activity of tryptamines such as 4-PrO-DMT modulates psychedelic-like effects and this may be relevant to clinical effects.
Psilocybin in conjunction with psychotherapy has received “Breakthrough Therapy” designation from the FDA for treating severe depression, but has not been approved by the FDA for the treatment of any medical condition and remains a DEA Schedule I controlled substance. Alternative psilocin prodrugs, such as psilacetin and 4-PrO-DMT potentially offer superior clinical and therapeutic properties to psilocybin. This recent collaborative research lays the groundwork for studying 4-PrO-DMT as a potential therapeutic and offers critical information on potential meaningful differences in the biological targets of various psychedelic tryptamines.